
BUSTILENE
Two clinical studies have demonstrated the efficiency of the product on painful symptoms of premenstrual mastodynia (pain, heaviness, edema…).
THE STUDY PERFORMED IN 2018
STYDY TITLE
Evaluation of the efficacy and tolerance of a medical device on the improvement of signs and symptoms of mastodynia during premenstrual syndrome – pain related to breast tension-.
CRO
DERMSCAN-Pharmascan, Villeurbanne –69100. France
SMO/Principal Investigator
Dr BALLAM, Gynaecologist. Insight Research – Quatre Bornes - Mauritius
STUDY DESIGN
Prospective, open, in before/after, mono centric on 44 subjects over18 years old
STUDY PROCEDURE
Total duration of the study: 5 days per patient. 2 visits :
- D0:inclusion
- D28:after one cycle
TREATMENT
To apply during the whole study, at least twice a day (on the morning and on the evening) and more if necessary, a nut of cream at least on each breast. The application was done by prolonged massage by effleurage of the cream on the skin.
PRIMARY ENDPOINT
The primary objective of the study was to evaluate the speed decrease of the pain related to breast tension, scored by subjects on VAS.
SECONDARIES ENDPOINTS
To evaluate the intensity of the pain related to breast tension, scored by the subjects on VAS.
To evaluate the speed of the breast tension decrease scored by the subjects on VAS.
To evaluate the safety and local tolerance of the tested MD.
To subjectively evaluate the acceptability and future use of the tested MD.
RESULTS/CONCLUSION
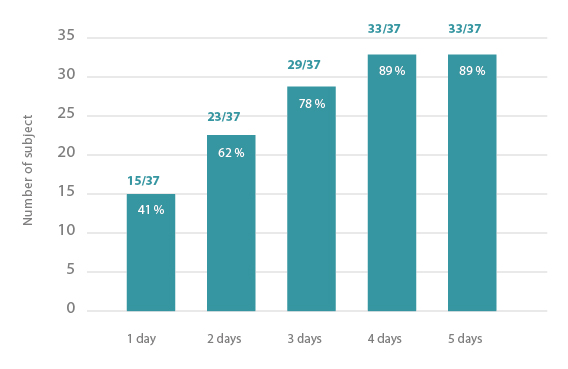
Speed of decrease of pain : At the first use of the device, the intensity of the pain linked to breast tension was of 7.7±0.7 on average. From D3, a significant decrease of the pain intensity was observed.
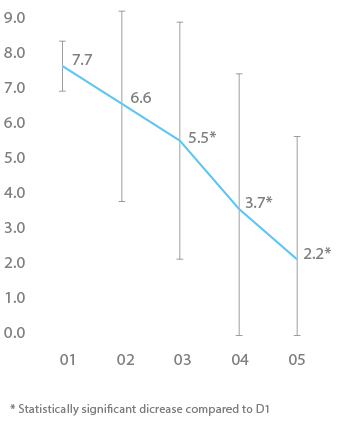
Decrease of pain intensity:
At the first use, the mean intensity was of 7.7±0.7. When a decrease (>0.5 cm on the VAS) was observed or otherwise when the device was stopped, the intensity of the pain was of 4.2±3.7, which represent a mean significant decrease of 3.5±3.4 (p<0.0001).
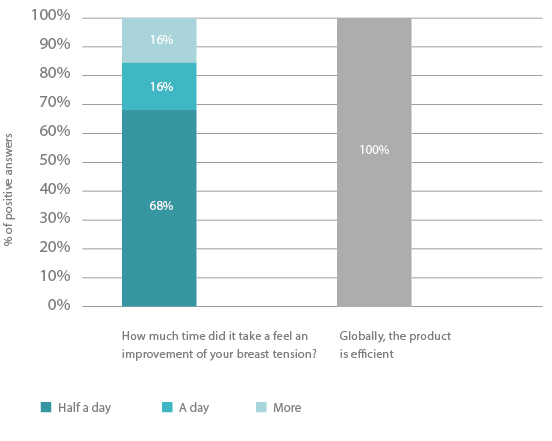
Intensity of breast tension sensations:
OVERALL CONCLUSION
The device tested can be considered as efficient on signs and symptoms of mastodynia. Indeed, a decrease of the pain linked to breast tension and of the breast tension sensations were reported by most subjects after two days of use of the device. Subjects very well appreciated the device for its efficacy, its properties and its acceptability. The device was also very well tolerated on the cutaneous level.
other clinical study
STUDY TITTLE
‘’Study of the clinical efficacy of Bustilène cream for local use, in the treatment of mastodynia and premenstrual syndrome’’
INVESTIGATORS
• Doctor Brigitte Clavier, Gynecological and obstetric Clinic – University Hospital Center Charles-Nicolle – Rouen, France
• Doctor Valérie Saubry-Bobet, Clinic du Cèdre - Bois-Guillaume, France
STUDY DESIGN
Open-label preliminary study on 45 women.
STUDY PROCEDURE
This clinical study was conducted over a period optimally corresponding to 3 menstrual cycles.
- Primary endpoint: Reduction of breast pain, tension, heaviness, feelings of heat and inflammation, reduction of oedema.
- Secondary endpoint: rapidity of action, durability, cosmetic evaluation of the product.
Each patient received the tested product.
The patient must applied a small amount of cream onto each breast, twice a day, for about a fortnight preceding the presumed date of menstrual bleeding.
- C0 – 1st visit : Inclusion
- C1 – 2nd visit was held during the premenstrual period, about one week before
the presumed date of the menstrual bleeding.
- C2 – 3rd visit was held also about one week before the presumed date of the fourth period
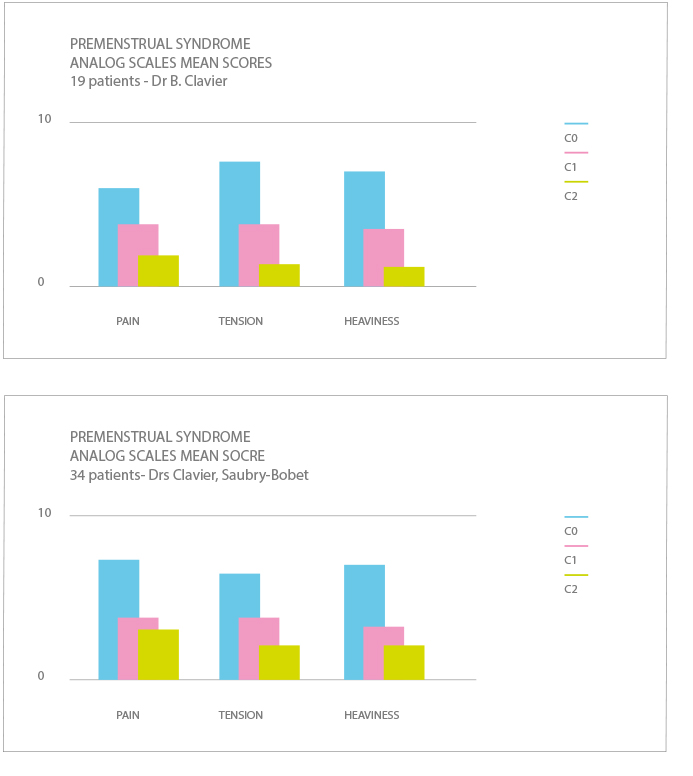
RÉSULTATS/CONCLUSIONS
Overall, between the inclusion visit C0 and the last visit C2 corresponding to an optimal treatment of 3 cycles, a marked improvement was observed for:
- Pain in 63% of cases
- Tension in 71% of cases
- Heaviness in 69% of cases
Results are identical on other parameters excepted for oedema where an improvement was observed in 100% of cases.
EXPERTS CONCLUSION
This study conducted in Rouen on a population of private practice patients and a population of patients attending the teaching hospital breast clinic demonstrated a number of interesting points:
• The diversity of the clinical symptoms and signs presented by the patients included.
• The very small number of patients who dropped out of the study for reasons related to the test product (texture, absence of improvement).
• The very rapid and indisputable efficacy of the product on subjective symptoms and the marked improvement of the breast examination performed by the clinician.
• Almost all patients (87%) were completely satisfied by the relief provided by the product and wanted to continue treatment.
